
Insulator, semiconductor, conductor
Solid-state materials can be classified into three groups: insulators, semiconductors and conductors. Insulators are materials having an electrical conductivity
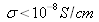 (like diamond: 10-14S/cm); semiconductors have a conductivity
(like diamond: 10-14S/cm); semiconductors have a conductivity
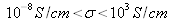 (for silicon it can range from 10-5S/cm to 103S/cm); at last conductors are materials with high conductivities :
(for silicon it can range from 10-5S/cm to 103S/cm); at last conductors are materials with high conductivities :
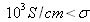 (like silver: 106S/cm.)
(like silver: 106S/cm.)
The electrical properties of a given material depend on the electronic populations of the different allowed bands. Electrical conduction is the result of electron motion within each band. When an electric field is applied to the material, electrons start to move in the direction opposed to the direction of the electric field. An empty energy band (in which there is no free electron) does not of course participate in the formation of an electric current. It is also the case for a fully occupied band. Indeed, an electron can move provided that, whenever it leaves its site, it can find some free space elsewhere (another available site within its energy band, called a “hole”), where it can go. A material with fully occupied or empty energy bands is then an insulator. This is the case when the gap energy exceeds ~9eV, because for such gaps, the thermal energy at 300K (~25 meV) is clearly insufficient to allow electrons from the valence band to be promoted to the conduction band. In this case the valence band (and all bands of lower energy) is fully occupied, and the conduction band is empty.
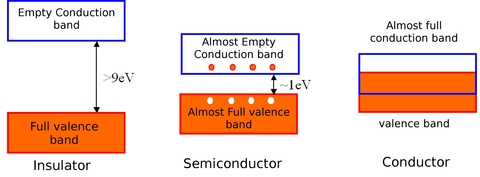
A semiconductor is primarily an insulator at 0K. However, since the energy gap is lower compared to insulators (~1eV), the valence band is slightly thermally populated at room temperature, whereas the conduction band is slightly depopulated. Since electrical conduction is directly connected to the number of electrons in the “almost empty” conduction band and to the number of holes in the “almost fully occupied” valence band, it can be expected that the electrical conductivity of such an intrinsic semiconductor will be very small.
For a conductor, conduction bands and valence bands are not separated and there is therefore no energy gap. The conduction band is then partially occupied (even at low temperatures), resulting in a “high” electrical conductivity.